Abstract
Background:FLT3 mutations (FLT3m) are detected in approximately 30% of newly diagnosed (ND) pts with AML and are associated with poor prognosis. FLT3m can also emerge in relapse in pts with previously wild-type FLT3 AML; however, their frequency and clinical implications are not well studied. Our objective was to identify pts who relapsed with emergent FLT3m and compare their clinical and pathological characteristics to those who relapse without FLT3m.
Methods: We identified 686 pts with ND AML treated at our institution between 12/2005 and 01/2020, who achieved complete remission (CR) / CR with incomplete count recovery (CRi) and subsequent relapse of AML. Pts with available FLT3 status (ITD and TKD) at diagnosis and within 4 weeks of first relapse were included (N=300). A multiplex fluorescent-based PCR analysis followed by capillary electrophoresis for detection of FLT3 ITD and/or TKD was performed on DNA isolated from BM aspirate samples. Next-generation sequencing (NGS) at baseline was performed using the Illumina MiSeq platform with multigene panels (either 28-gene, 53-gene, or 81-gene) interrogating genes recurrently mutated in myeloid neoplasms.
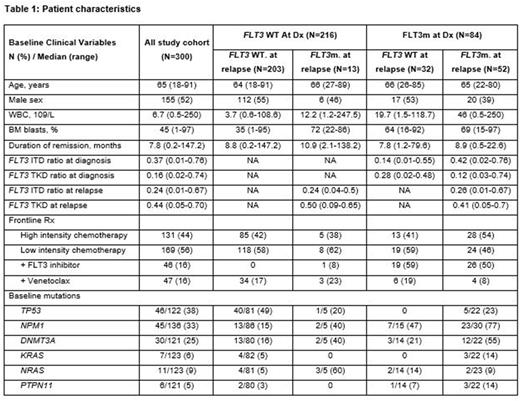
Results: Among 300 pts who achieved CR/CRi after induction and eventually relapsed, 216 (72%) had FLT3 wild-type (WT) AML and 84 (28%) had FLT3m AML at diagnosis. Baseline clinical features are shown in Table 1. 43 (14%) pts had a change of FLT3 status between baseline and relapse: 30 pts had FLT3m at baseline that wasn't detected at relapse, and 13 pts with FLT3 WT AML at baseline experienced relapse with FLT3m AML.
The median remission duration (until 1st relapse) for the entire study cohort pts withFLT3 WT and pts with FLT3m AML was 7.8 months (range, 0.2-147.2), 9 months (0.2-147.2), and 6.1 months (range, 0.5-79.6), respectively.
FLT3 wild-type at Diagnosis
Among 216 pts with FLT3 WT AML at baseline, 203 (94%) and 13 (6%) experienced relapse with FLT3 wild-type and FLT3m AML, respectively. Comparison of baseline molecular features of FLT3 WT and FLT3m cohorts showed that pts who had relapsed AML with a positive FLT3m had more frequent concomitant NRAS mutations compared with pts who had FLT3 WT AML at relapse (60% vs 5%; p=0.003). No significant difference was seen in the frequency of TP53 (49% vs. 20%; p=0.36), NPM1 (15% vs. 40%; p=0.19), DNMT3A (16% vs. 40%; p=0.21), KRAS (5% vs. 0%; p=1), or PTPN11 (3% v. 0%; p=1) mutations between the two cohorts, respectively.
Among 13 pts with AML with emergent FLT3m, 12 had diploid cytogenetics at diagnosis, but only 6 maintained the diploid status at relapse. The remaining 6 pts developed karyotypic clonal evolution at relapse (complex, n=1; KMT2A rearrangement, n=1; trisomy 8, n=1; other alterations, n=3). We had paired baseline and relapse NGS performed in 5 of 13 pts with relapsed AML, among these AMLs acquired new mutations. Nine newly acquired mutations were detected in the following genes: NPM1 (n=1), KRAS (n=1), RUNX1 (n=1), IDH2 (n=1), DNMT3A (n=1), ZRSR2 (n=1), WT1 (n=2), and STAG2 (n=1). Two of 5 pts had AML with no mutations at baseline, and 1 pt had AML with the same mutation profile at baseline and relapse.
The median OS after relapse was 5.3 months and 5.5 months in pts with WT vs. FLT3m AML, respectively (P=0.29) (Figure 1).
FLT3 mutant at diagnosis
Among 84 pts with FLT3m AML at diagnosis, 52 (62%) and 32 (38%) pts experienced relapse with FLT3m and WT AML, respectively. Comparison of baseline molecular features of FLT3 WT and FLT3m AML showed a trend towards a higher frequency of NPM1 (77% vs. 47%; p=0.09) and DNMT3A mutations (55% vs. 21%; p=0.08) in patients who experienced relapse with FLT3m AML compared with FLT3 WT AML. No difference in the frequency TP53 (0% vs. 23%; p=0.13), KRAS (0% vs. 14%; p=0.27), NRAS (14% vs. 9%; p=0.63) and PTPN11 (7% vs. 14%; p=1) mutations was observed among both cohorts, respectively.
The median OS after relapse was 8.1 months vs. 3.9 months in pts with FLT3 WT vs. FLT3m AML, respectively (p=0.005) (Figure 2).
Conclusion: Our data suggest that FLT3m are more likely to emerge at AML relapse with concomitant NRAS mutations and indicate that FLT3m testing should be performed at relapse, irrespective of FLT3status at diagnosis. It is common for the FLT3 status to change, either from mutant to WT or vice versa. FLT3 status has immediate and clinically important implications for selection of appropriate therapeutic agents in the setting of salvage therapy.
Disclosures
Kanagal-Shamanna:Amgen: Consultancy; Novartis: Consultancy; Aptitude Health: Speakers Bureau; Physicians Education Resource: Speakers Bureau. Loghavi:Amgen: Research Funding; Abbvie: Consultancy, Current equity holder in publicly-traded company; GLG: Consultancy; Astellas: Research Funding; QualWorld: Consultancy; PeerView: Honoraria. Daver:Agios, Celgene, SOBI and STAR Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees; Kartos and Jazz Pharmaceuticals: Other: Data monitoring committee member; Karyopham Therapeutics and Newave Pharmaceutical: Research Funding; Astellas, AbbVie, Genentech, Daiichi-Sankyo, Novartis, Jazz, Amgen, Servier, Karyopharm, Trovagene, Trillium, Syndax, Gilead, Pfizer, Bristol Myers Squibb, Kite, Actinium, Arog, Immunogen, Arcellx, and Shattuck: Consultancy, Other: Advisory Role; Astellas, AbbVie, Genentech, Daiichi-Sankyo, Gilead, Immunogen, Pfizer, Bristol Myers Squibb, Trovagene, Servier, Novimmune, Incyte, Hanmi, Fate, Amgen, Kite, Novartis, Astex, KAHR, Shattuck, Sobi, Glycomimetics, Trillium: Research Funding. Jabbour:Pfizer: Other: Advisory Role, Research Funding; Genentech: Other: Advisory Role, Research Funding; Bristol Myers Squibb: Other: Advisory Role, Research Funding; Takeda: Other: Advisory Role, Research Funding; Amgen: Other: Advisory Role, Research Funding; Adaptive Biotechnologies: Other: Advisory Role, Research Funding; AbbVie: Other: Advisory Role, Research Funding; Spectrum: Research Funding. Kadia:Iterion: Research Funding; Astex: Honoraria; Astellas: Research Funding; AstraZeneca: Research Funding; Genfleet: Research Funding; Amgen: Research Funding; Delta-Fly: Research Funding; Genentech: Consultancy, Research Funding; Servier: Consultancy; Pfizer: Research Funding; Ascentage: Research Funding; cyclacel: Research Funding; PinotBio: Consultancy; Glycomimetics: Research Funding; Novartis: Consultancy; cellenkos: Research Funding; Regeneron: Research Funding; BMS: Consultancy, Research Funding; JAZZ: Consultancy, Research Funding; Agios: Consultancy; Abbvie: Consultancy, Research Funding. Short:AstraZeneca: Consultancy; Takeda Oncology: Consultancy, Research Funding; Astellas: Research Funding; Amgen: Consultancy, Honoraria; Pfizer: Consultancy; Stemline Therapeutics: Research Funding; Novartis: Consultancy. DiNardo:LOXO: Research Funding; Notable Labs: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria; Jazz: Honoraria; Forma: Research Funding; Foghorn: Honoraria, Research Funding; Bristol Myers Squibb: Honoraria, Research Funding; Gilead: Honoraria; Kura: Honoraria, Membership on an entity's Board of Directors or advisory committees; ImmuneOnc: Honoraria, Research Funding; GenMab: Membership on an entity's Board of Directors or advisory committees; GlaxoSmithKline: Membership on an entity's Board of Directors or advisory committees; Cleave: Research Funding; Astex: Research Funding; Takeda: Honoraria; Bluebird Bio: Honoraria; Astellas: Honoraria; Servier: Consultancy, Honoraria, Research Funding; AbbVie: Consultancy, Research Funding. Borthakur:Catamaran Bio, Abbvie, PPD Development, Protagonist Therapeutics, Janssen: Consultancy; Astex Pharmaceuticals, Ryvu, PTC Therapeutics: Research Funding; Pacylex, Novartis, Cytomx, Bio Ascend: Membership on an entity's Board of Directors or advisory committees. Pemmaraju:stemline: Consultancy; abbvie: Consultancy; immunogen: Consultancy; mustangbio: Research Funding; incyte: Consultancy; novartis: Research Funding; pacylex: Consultancy, Research Funding; samus: Research Funding; daiichi sankyo: Research Funding; cellectis: Research Funding; cellularity: Research Funding. Garcia-Manero:BMS: Consultancy, Honoraria, Research Funding; Curis: Honoraria, Research Funding; Aprea: Honoraria; Novartis: Honoraria, Research Funding; Genentech: Honoraria, Research Funding; Acceleron Pharma: Consultancy; AbbVie: Honoraria, Research Funding; Gilead Sciences: Research Funding; Astex: Consultancy, Honoraria, Research Funding. Ravandi:Syos: Consultancy, Honoraria, Research Funding; AstraZeneca: Consultancy; Astellas: Consultancy, Honoraria, Research Funding; Abbvie: Consultancy, Honoraria, Research Funding; BMS/Celgene: Consultancy, Honoraria, Research Funding; Astex/Taiho: Membership on an entity's Board of Directors or advisory committees, Research Funding; Biomea Fusion, Inc.: Research Funding; Amgen: Honoraria, Research Funding; Novartis: Consultancy; Prelude: Research Funding; Xencor: Research Funding; Amgen: Honoraria, Research Funding. Kantarjian:Astellas Health: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria, Research Funding; Ascentage: Membership on an entity's Board of Directors or advisory committees, Research Funding; Daiichi-Sankyo: Consultancy, Research Funding; AbbVie: Honoraria, Research Funding; ImmunoGen: Research Funding; Novartis: Honoraria, Research Funding; KAHR Medical Ltd: Honoraria, Membership on an entity's Board of Directors or advisory committees; Jazz Pharmaceuticals: Research Funding; Ipsen Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees; NOVA Research: Honoraria; Pfizer: Honoraria, Research Funding; Takeda: Honoraria. Yilmaz:Pfizer: Research Funding; Daiichi-Sankyo: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal